C1.7.2.2 Analysis of Triprotic Phosphoric Acid by Titration Leybold LD Didactic
$ 6.725,00 Original price was: $ 6.725,00.$ 1.681,25Current price is: $ 1.681,25.
Analysis of Triprotic Phosphoric Acid by Titration
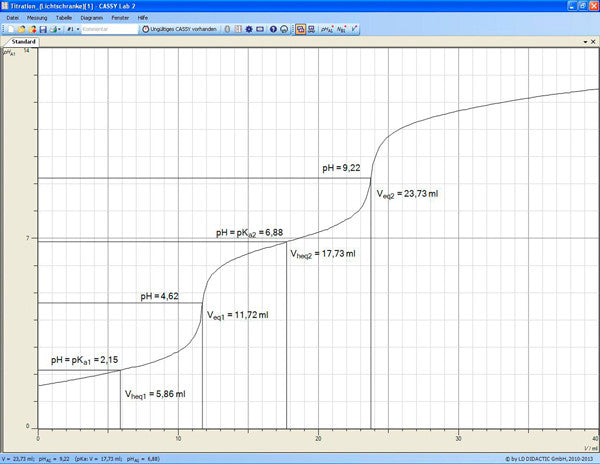
Phosphoric acid is a triprotic acid. When dissolved in water, it first gives up one proton and dissociates to dihydrogen phosphate, i.e. it reacts like a monoprotic acid (see formula 1). The addition of sodium hydroxide, e.g. during titration, first leads to complete dissociation of the phosphoric acid into dihydrogen phosphate.
The second protolysis, i.e. the reaction to hydrogen phosphate (see formula 2), occurs only after a high pH value has been reached, approx. pH 9. The third protolysis requires a considerably higher pH value (see formula 3). In experiment C1.7.2.2, the first two protolysis steps of phosphoric acid are determined in an automatic titration. 
Prompt shipping and expert packing
Our long-standing relationship with UPS FedEx DHL and other global carriers lets us offer a range of shipping services. Our warehouse personnel will pack all goods to our exacting specifications. Your items are carefully inspected and secured properly prior to shipping. We deliver to thousands of clients every day from all over the world. This demonstrates our dedication to become the biggest online retailer in the world. Both Europe and the USA have warehouses and distribution centres.
Note: Orders with more than one product are assigned a particular processing time based on the individual product.
We will thoroughly inspect all products before they are shipped. The majority of orders today are shipped in 48 hours. Expected delivery times are between 3 and 7 days.
Returns
The inventory is constantly changing and we cannot control it completely because of the fact that multiple parties are involved, which includes our factory and warehouse. The actual stock levels can fluctuate at any point. It's possible that you may not receive your order once it has been placed.
The policy is for 30 days. If you haven't received the product within the 30 days period, we are not able to issue the option of a refund or exchange.
You are able to return a product when it's unopened and in the same condition as when you received it. It must also be returned in its original packaging.
Related products
Science & Laboratory
200313 LeyLab 365 Digital Content for Science Teaching Annual License Leybold LD Didactic
Science & Laboratory
Science & Laboratory
Science & Laboratory
204703 Basic Science Kits- Biology 3/3 (Microscopy / Genetics) Leybold LD Didactic
Science & Laboratory
204405 Basic Science Kit Physics – Optics Leybold LD Didactic
Science & Laboratory
Science & Laboratory
204690EN LIT: Basic Science Kit Chemistry Leybold LD Didactic
Science & Laboratory
Science & Laboratory
Science & Laboratory
Science & Laboratory
16115 Mini Kit Magnetism/Electrostatics CORNELSEN-EXPERIMENTA
Science & Laboratory
204490EN LITERATURE: Basic Science Kit Physics Leybold LD Didactic
Science & Laboratory
Science & Laboratory
204606 Basic Science Kit Chemistry – Distillation Leybold LD Didactic
Science & Laboratory
204604 Basic Science Kit Chemistry – Physical Chemistry Leybold LD Didactic
Science & Laboratory
Science & Laboratory
Science & Laboratory
314201 Leybold Precision Dynamometer, 100.0 N Leybold LD Didactic
Science & Laboratory
Science & Laboratory
204403 Basic Science Kit Physics – Hydrostatics/Heat Leybold LD Didactic
Science & Laboratory
21-0602-R Laser Optical Set LOS1, Red Laser with Magnet Table KVANT
Science & Laboratory
204603 Basic Science Kit Chemistry – General Chemistry Leybold LD Didactic
Science & Laboratory
Science & Laboratory
Science & Laboratory
204702 Basic Science Kits- Biology 2/3 (Principles) Leybold LD Didactic
Science & Laboratory
204406 Basics Science Kit Physics Renewable Energy Leybold LD Didactic
Science & Laboratory
204601 Basic Science Kit Chemistry – Stand Material Leybold LD Didactic
Science & Laboratory
200310 LeyLab Digital Content for Science Teaching Leybold LD Didactic
Science & Laboratory
Science & Laboratory
Science & Laboratory
Science & Laboratory